Join Clinicians Worldwide
Evidence-based insights to enhance hearing care—twice a month
Subscribe Now
Evidence-based insights to enhance hearing care—twice a month
Subscribe NowMED-EL
Published Dec 06, 2017

The MED-EL Surgical Video Library offers complete surgical case studies from leading ENT surgeons. Created in cooperation with ARRI, these ultra-high-resolution videos capture precise movements and detailed structures with incredible clarity. Access is free and the easy-to-use library is optimized for desktop or mobile viewing.
Today, we have an excellent VIBRANT SOUNDBRIDGE Middle Ear Implant surgical case study to share. This video case study is part of our MED-EL Surgical Video Library, so it’s presented in ultra-high definition. With 4K resolution, you’ll have a clear view of surgical techniques and anatomical structures, including the posterior tympanotomy, exposure of the incus body, and attaching the Incus-SP-Coupler to the body and short process of the incus.
In this case study, Prof. Dr. Robert Mlynski demonstrates his surgical technique for right side implantation with the VIBRANT SOUNDBRIDGE VORP 503 Middle Ear Implant. Prof. Mlynski is the head of the ENT Department, Head and Neck Surgery, of the Otto Körner Clinic at the Rostock University Medical Center.
In this case, the 50-year-old female patient has bilateral congenital sensorineural hearing loss. This patient has already been using a VIBRANT SOUNDBRIDGE in her left ear since 2001, which she continues to benefit from.
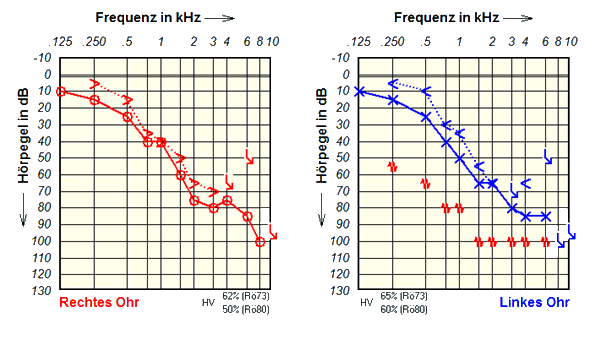
The pre-operative audiogram shows this patient has bilateral high-frequency sensorineural hearing loss.
As you can see from the audiogram, this patient has bilateral high-frequency hearing loss. Her hearing loss slopes from a mild hearing loss in the low frequencies to severe hearing loss in the high frequencies.
This patient could potentially be a candidate for EAS, but she has chronic otitis externa, which precludes the use of a hearing aid or acoustic element in her ear canal. As this patient was benefiting from her contralateral VIBRANT SOUNDBRIDGE, Prof. Mlynski chooses to also use a VSB for her right ear.

VIBRANT SOUNDBRIDGE VORP 503 Middle Ear Implant and SAMBA Audio Processor with magnified FMT and Vibroplasty couplers.
VIBRANT SOUNDBRIDGE is an ideal treatment option for mild-to-severe sensorineural hearing loss, as well as conductive and mixed hearing loss. It uses a Floating Mass Transducer (FMT) to directly stimulate middle ear structures with amplified sound vibrations. Depending on the hearing loss the FMT is attached to different structures of the middle ear using specialized Vibroplasty couplers.
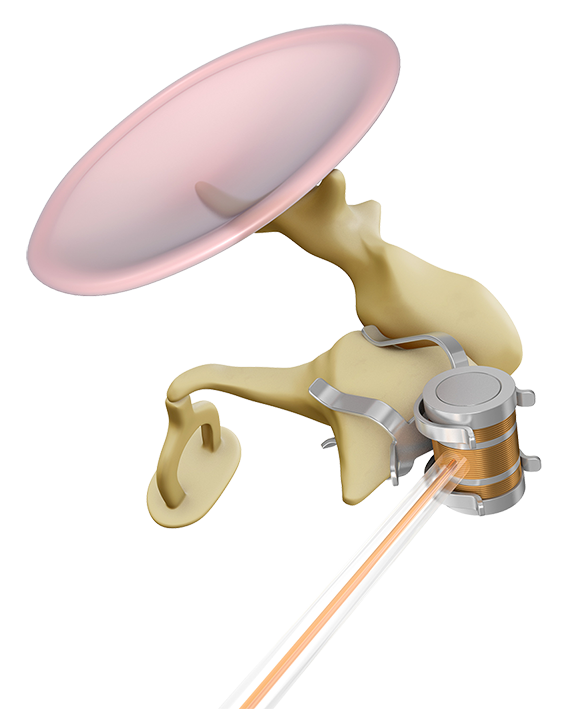
The Incus-SP-Coupler attaches the FMT to the body and short process of the incus, allowing simple connection through a posterior epitympanotomy. A posterior tympanotomy is not necessary, allowing surgeons to avoid the facial nerve and chorda tympani.
Watch now: Prof. Dr. Robert Mlynski demonstrates his VIBRANT SOUNDBRIDGE surgical techniques in this ultra HD video middle ear implant case study. (13 minutes)
Highlights to watch for in this middle ear implant surgical case study:
Want more surgical case studies like this? Check out the MED-EL Surgical Video Library for access to all our video case studies!
Have a question about using VIBRANT SOUNDBRIDGE for your patients? Let us know with our simple contact form.
*Not all products, indications, and features shown are available in all areas. Please contact your local MED-EL representative for more information.
MED-EL
Was this article helpful?
Thanks for your feedback.
Sign up for newsletter below for more.
Thanks for your feedback.
Please leave your message below.
CTA Form Success Message
Send us a message
Field is required
John Doe
Field is required
name@mail.com
Field is required
What do you think?
The content on this website is for general informational purposes only and should not be taken as medical advice. Please contact your doctor or hearing specialist to learn what type of hearing solution is suitable for your specific needs. Not all products, features, or indications shown are approved in all countries.
MED-EL
Get the latest research and resources to help people with every kind of hearing loss. Subscribe to the MED-EL Professionals Blog now.
Registration was successful
We’re the world’s leading hearing implant company, on a mission to help people with hearing loss experience the joy of sound.
Find your local MED-EL team
The content on this website is for general informational purposes only and should not be taken as medical advice. Please contact your doctor or hearing specialist to learn what type of hearing solution is suitable for your specific needs. Not all products, features, or indications shown are approved in all countries.