MED-EL
Published Sep 01, 2021
Thirty Years of Translational Research Behind MED-EL

Translating scientific research in laboratories into life-changing treatments for hearing loss is no miracle. Although cochlear implants are perceived as the first successful replacement of a human sense, advancements in cochlear implant technology are the result of decades of deliberate and diligent collaboration. For over three decades now, clinicians, their patients, and teams of specialists and researchers working with MED-EL have remained focused on one mission: overcoming hearing loss as a barrier to communication.
Yet behind each of MED-EL’s many hearing loss solutions is a story—and each of these stories begins with a patient or group of patients that inspired a research inquiry to address a specific condition that caused their hearing loss. From the moment clinicians express the need for a new solution, MED-EL’s aim is to provide these patients with the safest and most effective product to improve their daily lives.
Thirty Years of Translational Research Behind MED-EL is a collection of stories that reveal how enduring partnerships between clinicians and researchers have led to the development of MED-EL’s many innovations—all led by Ingeborg Hochmair and building upon her and Erwin Hochmair’s groundbreaking work on the world’s first microelectronic multi-channel cochlear implant that was successfully implanted in Vienna in 1977.
“What cochlear implants can do today seems like a miracle to many people. But this is actually the result of hard, painstaking, and collaborative work by teams of experts over the course of many years. After all, plenty of patience and stamina are required to persevere from the first idea or fundamental research to approval and reimbursement of costs for innovations in the field of cochlear implants. This is why I am delighted to pay homage to all those research collaborators and internal MED-EL colleagues with the compendium Thirty Years of Translational Research Behind MED-EL—they have supported us in pursuing our vision since 1990,” Dr. Ingeborg Hochmair, Founder and CEO of MED-EL.
The Stories Behind MED-EL’s Life-Changing Devices
The publication honors approximately 200 external scientific collaborators who have contributed to the achievement of milestones, the development of evidence, and, ultimately, the creation of approved, refundable products and solutions for people of all age groups with hearing loss.
Each product or solution began as an initial idea or need expressed by clinicians as a treatment option for a patient (or patients) with specific indications, such as those:
- Born with severe-to-profound bilateral hearing loss (Chapter 1)
- Profoundly deaf in the high-frequency region but not in the low-frequency region (Chapter 2)
- With a nonfunctional auditory nerve (Chapter 3)
- With single-sided deafness (Chapter 4)
- With congenital abnormal inner ear malformations or special cochlear needs (Chapter 7)
Broader innovations in cochlear implant technology are also covered in the compendium including:
- Advancements in signal processing and audio processors (Chapter 5)
- Drug delivery in cochlear implantation to improve preservation of residual hearing (Chapter 6)
- Ingeborg Hochmair’s views on the overall goals, learnings, and successes of translational research in hearing loss treatment (Chapter 8)
“All of us at MED-EL are very proud of the pioneering technological innovations we have developed over the past thirty years thanks to close dialogue with medical experts, scientists, and implant users. Together with our collaborators around the world, we will continue working towards our vision of facilitating hearing that is as natural as possible for all those with hearing loss,” Dr. Ingeborg Hochmair.
Translational Research: Five Categories of Cochlear Implants
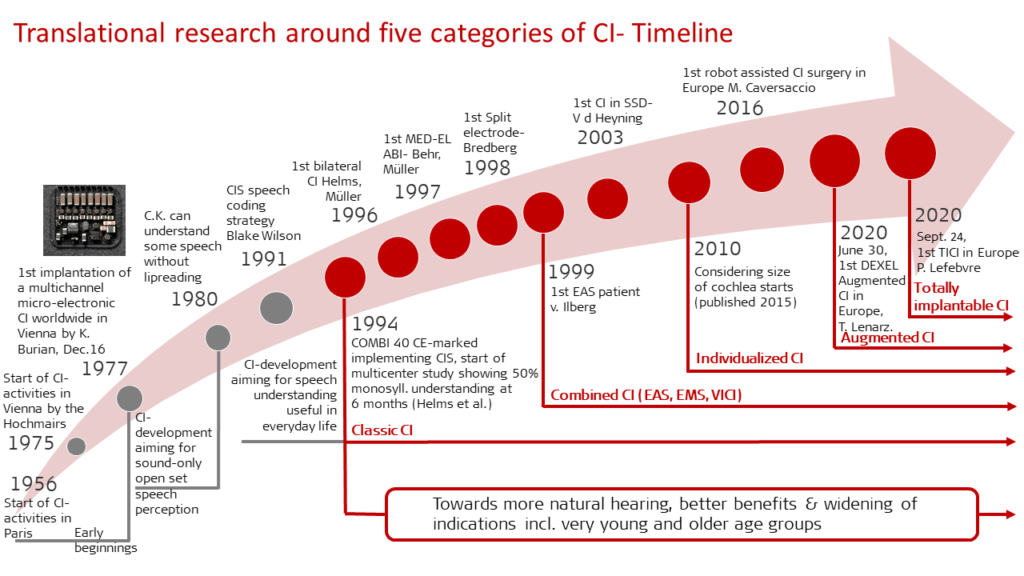
In the final chapter of the compendium, Dr. Ingeborg Hochmair reflects on key milestones reached in the development of five categories of cochlear implants—all of which are based on early cochlear implants and currently undergoing further development.
Classic Cochlear Implants
Since 1994 developments related to classic cochlear implants have been focused on making CIs sound more natural, improving outcomes for recipients with SSD, bilateral or asymmetric hearing loss, widening indications, enhancing hearing preservation and connectivity, as well as remote care and bimodal stimulation (when recipients use a hearing aid in the contralateral ear).
Combined Cochlear Implants
This includes cochlear implants combined with other types of stimulation such as EAS (electric stimulation with acoustic amplification), EMS (electromechanical stimulation via a CI with a vibratory actuator), and VICI (a CI that also electrically stimulates the vestibular organ in loss of vestibular function).
Individualized Cochlear Implants
Cochlear implants from MED-EL are currently routinely individualized via surgical planning software that assists in selecting the optimal length electrode and postoperative anatomy-based fitting as well as supporting hearing and structure preservation.
Augmented Cochlear Implants
Augmented cochlear implants combines cochlear implantation with the release of a substance into the cochlea preoperatively, intraoperatively, postoperatively or eluted from the electrode over a period of time. For example, corticosteroids can be applied during cochlear implantation to minimize intra-cochlear fibrous tissue formation around the electrode.
Totally Implantable Cochlear Implants
The first implantation of a totally implantable cochlear implant (TICI) in Europe took place in September 2020 as part of a feasibility study. The TICI includes a rechargeable battery, microphone and audio processor all in one unit, which is implanted so that the system no longer requires any external components to be worn on the skin.
Each of these five fields of cochlear implants represent distinct areas in which future innovations will further improve quality of life and hearing outcomes for recipients.
Deeper Insights for Hearing Professionals
The compendium Thirty Years of Translational Research Behind MED-EL provides hearing professionals rare and unique insights into the milestones from thirty years of research partnerships concerning cochlear and brainstem implants (CI and ABI).
External scientists, audiologists, surgeons, and rehabilitation professionals played key roles in collaborating with MED-EL to collect evidence, and, ultimately, help develop approved, refundable products and solutions for everyday use by people of all age groups with hearing loss.
Audiologists
Audiologists, in particular, might want to check out:
- Chapter 4 on “CI in Single-Sided Deafness” to see the key role that audiologists and clinicians played in helping to expand CI as a treatment option from adults with unilateral tinnitus in SSD to those with the indication of SSD and asymmetric hearing loss (AHL) in adults and children—and how MED-EL CIs were the first to be approved for patients over the age of five with SSD and SHL by the US FDA
- Chapter 2 to discover more about the optimized fitting procedure in EAS patients so that electrical stimulation and acoustic stimulation do not overlap.
- Chapter 1, which discusses simultaneous versus sequential CI implantation
- Chapter 5, which covers developments related to signal processing in audio processors as well as the latest technology for bimodal hearing
Surgeons
Some highlights that ENT surgeons and otologists won’t want to miss are:
- Chapter 7, which describes how MED-EL electrodes have been designed and produced to accommodate the cochlear needs of individuals with special and challenging cochlear conditions.
- Chapter 6, which explores the use of the intra-cochlear catheter to administer drug substances into the cochlea to minimize the intra-cochlear fibrous tissue formation around the electrode.
- Chapter 3, which reveals the fascinating story behind the development of MED-EL’s auditory brainstem implant. This section is also highly recommended for experienced neurosurgeons.
Rehabilitation Specialists
Rehabilitation specialists may benefit, in particular, from insights found in:
- Chapter 1, which provides insights into how rehabilitation specialists can help recipients get used to the second cochlear implant when it is implanted sequentially and may require some adjustments to match with the first cochlear implant
- Chapter 3, which describes the crucial role of rehabilitation for patients with auditory brainstem implants
A Freely Accessible Publication to Celebrate Thirty Years of Collaboration
Aspiring scientists and the future generation of specialists can take inspiration from Thirty Years of Translational Research Behind MED-EL. The insightful collection of translational research success stories appears as a supplement to the medical journal Acta Oto-Laryngologica by publishing house Taylor & Francis and can be downloaded for free.
The full publication is available to read online here.
Special thanks to Dr. Anandhan Dhanasingh, Head of Translational Science Communication, R&D, at MED-EL for contributing to this article.
MED-EL
Was this article helpful?
Thanks for your feedback.
Sign up for newsletter below for more.
Thanks for your feedback.
Please leave your message below.
CTA Form Success Message
Send us a message
Field is required
John Doe
Field is required
name@mail.com
Field is required
What do you think?
MED-EL



