Join Clinicians Worldwide
Evidence-based insights to enhance hearing care—twice a month
Subscribe Now
Evidence-based insights to enhance hearing care—twice a month
Subscribe NowMED-EL
Published Jun 14, 2017
In the first part of this series on cochlear implant sound quality, we looked at electrode arrays. Specifically, we saw how long electrode arrays are necessary for place-pitch match in the mid-to-low frequencies. If an array can’t reach the second turn of the cochlea, it can’t provide natural tonotopic coding for lower frequencies.
Yet beyond the first turn, place-pitch match is only the first step. Now we’ll look at the second half of the equation—sound frequency-matched rate coding in the apical region.
Let’s take a look at why you need a sound coding strategy that provides both tonotopic (place) and temporal (rate) coding for the most accurate pitch perception.
With natural hearing, a more complex arrangement comes into play in the second turn of the cochlea. In addition to tonotopic coding, a form of rate coding appears. Here in the apical region, hair cells are able to trigger nerve signals in sync with the frequency of sound signals.1,2
For mid-to-low frequencies, each cycle of a sound wave opens and closes a hair cell response—like turning a light switch on and off. Sound frequency is measured in cycles per second (Hz), so in theory, a 110 Hz sound wave triggers 110 compound action potentials per second in the auditory nerve fiber. A 263 Hz tone would trigger 263 compound action potentials per second, a 440 Hz tone would trigger 440 compound action potentials, and so on. This phase-locking synchronizes the action potentials to the incoming sound wave.2
This one-to-one rate coding enables precise natural frequency tuning that likely wouldn’t be possible with place coding alone. It’s often referred to as temporal coding, because it can provide the precise time coding of sound. Temporal coding is believed to have an important role in speech understanding in noise, pitch perception, music appreciation, and sound localization.1,2
At approximately 1,000 Hz, the firing rate of individual nerve fibers becomes increasingly saturated and tonotopic coding takes over. That’s why tonotopic coding and temporal coding are both essential in natural hearing.1,2
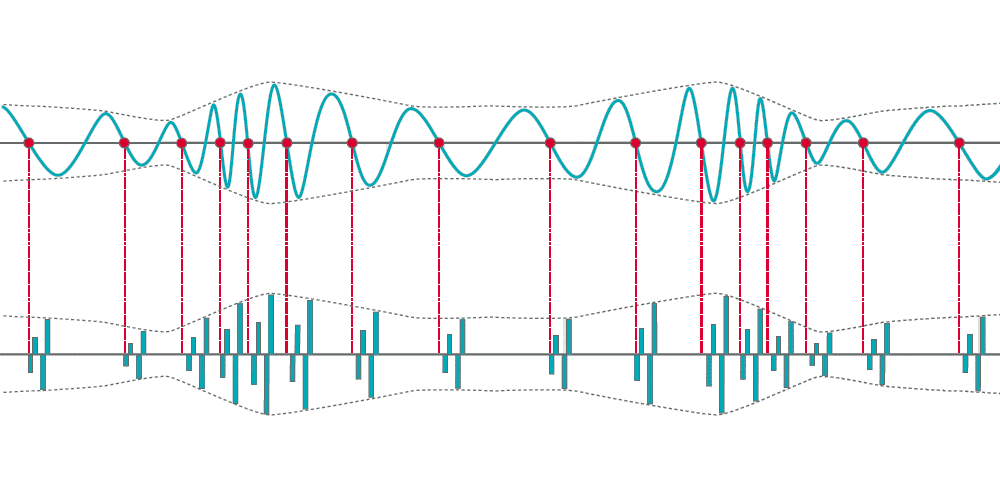
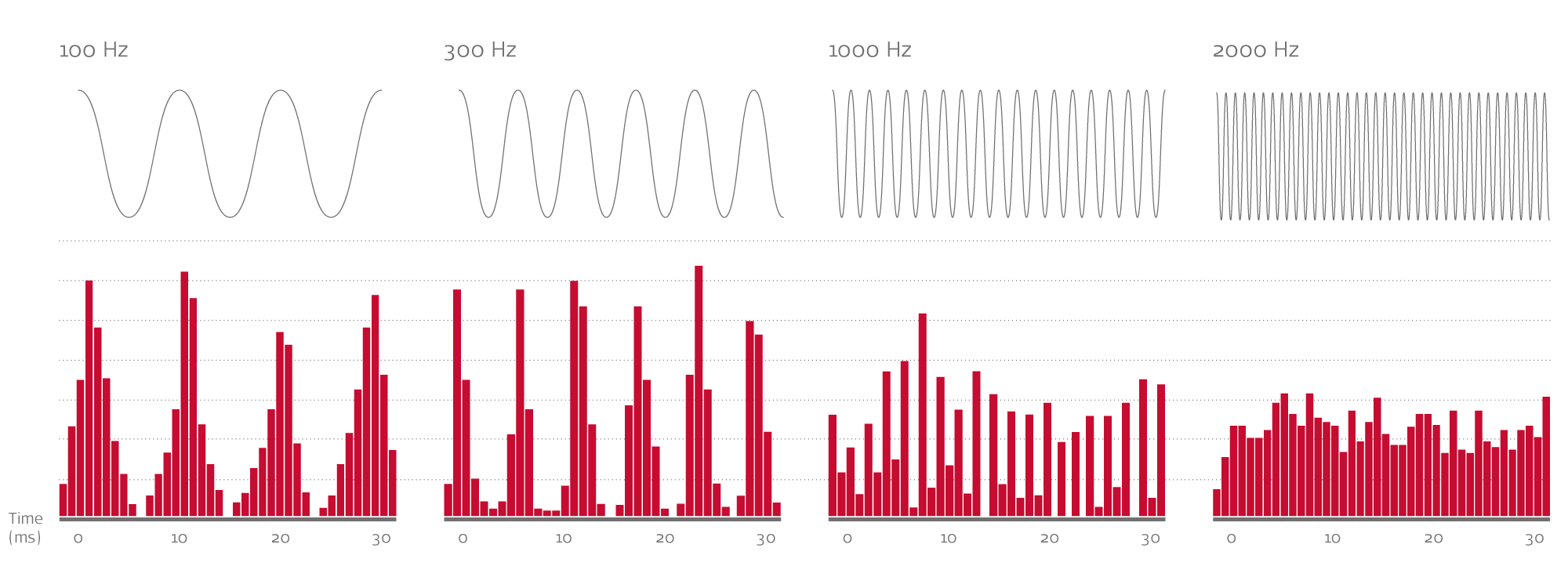 Phase-locked temporal coding in the auditory nerve can be seen up to roughly 1000 Hz, where the individual neural firing rate becomes saturated. Visual approximation of phase-locking; image for illustrative purposes only, based on Pickles, et al. (2013).
Phase-locked temporal coding in the auditory nerve can be seen up to roughly 1000 Hz, where the individual neural firing rate becomes saturated. Visual approximation of phase-locking; image for illustrative purposes only, based on Pickles, et al. (2013).
If a long array can provide place-pitch match in the second turn of the cochlea, why does rate coding matter for cochlear implant recipients?
Accurate rate coding in the second turn of the cochlea is essential for more natural perception of mid-to-low frequencies. If the implant uses a fixed rapid pulse rate (e.g. 1,500 pulses per second), it can distort pitch perception on electrodes in the apical region. A pulse rate that’s too high will cause pitch perception to shift upwards. 3,4,5
Think of it like playing a record on a turntable. At the right speed, the music sounds natural. But if you speed up the turntable, it shifts everything up in pitch. So, for accurate low-frequency perception, an implant needs to mimic natural rate coding in the second turn of the cochlea. This means slowing down pulse rate to match the sound frequency.3,4,5,6
Let’s look at recent pitch-matching experiments with recipients who had relatively normal hearing in their non-implanted ear, or even residual hearing in their implanted ear. These studies have shown rate-matched pulses on place-matched electrode—for example, 100 pps for a 100 Hz signal on an apical electrode in the second turn—can deliver accurate pitch perception down to approximately 100 Hz.3,4,6,7
These low frequencies are essential for a rich, full, resonant sound—without the low frequencies, sound quality can be “robotic”, “tinny”, “echoey”, or “mechanical”.8
However, rate coding isn’t effective in the first turn of the cochlea—a low pulse rate on a short array can’t effectively lower the pitch enough, and can produce an unpleasant or annoying sound quality.3,5,6 So if an electrode array can’t reach the apical region, it can’t mimic natural rate coding. This means low-to-mid pitches cannot be mapped according to the Greenwood function. Without access to accurate tonotopic or rate coding, natural perception of the lower frequencies is out of reach on short arrays.3,4,7,9,10
There’s only one way to achieve more natural pitch perception across the whole cochlea—by combining long electrode arrays with precise, rate-matched stimulation.4,5,6,7
What benefits can long arrays and pitch-matched rate coding offer for your patients?
And lastly, “An unparalleled restoration of tonotopic pitch perception”—Rader et al. (2016)4
Have a question about long arrays and rate-matched sound coding? Leave a comment below!
We’ll be taking a closer look at sound coding in upcoming articles—subscribe now to make sure you don’t miss any posts!
*Not all products, indications, and features shown are available in all areas. Please contact your local MED-EL representative for more information.
References:
MED-EL
Was this article helpful?
Thanks for your feedback.
Sign up for newsletter below for more.
Thanks for your feedback.
Please leave your message below.
CTA Form Success Message
Send us a message
Field is required
John Doe
Field is required
name@mail.com
Field is required
What do you think?
The content on this website is for general informational purposes only and should not be taken as medical advice. Please contact your doctor or hearing specialist to learn what type of hearing solution is suitable for your specific needs. Not all products, features, or indications shown are approved in all countries.
MED-EL
Get the latest research and resources to help people with every kind of hearing loss. Subscribe to the MED-EL Professionals Blog now.
Registration was successful
We’re the world’s leading hearing implant company, on a mission to help people with hearing loss experience the joy of sound.
Find your local MED-EL team
The content on this website is for general informational purposes only and should not be taken as medical advice. Please contact your doctor or hearing specialist to learn what type of hearing solution is suitable for your specific needs. Not all products, features, or indications shown are approved in all countries.