
Nancy M. Young, MD, FACS, FAAP
Published Oct 12, 2020
Importance of Safe MRI Access in Cochlear Implants: Dr. Nancy M. Young
Dr. Nancy Young shares her extensive experience and research on safe MRI access with cochlear implants.

MRI is an essential tool in modern medicine. Our priority has always been to offer safe, comfortable access to MRI scans for our cochlear implant recipients. With a secure magnet design, there has never been a report of magnet dislocation in a MED-EL cochlear implant, even going back more than 25 years.
In 2015, we pushed this safety even further by introducing the SYNCHRONY cochlear implant. SYNCHRONY has a revolutionary magnet design—a rotating, self-aligning diametric magnet in a secure titanium housing. For the first time, cochlear implants could undergo 3.0 Tesla MRI without any need for surgery (unless required for diagnostic reasons) and without any risk of magnet dislocation. SYNCHRONY effectively eliminates noticeable torque forces, so even a head bandage or splint kit is not needed.**
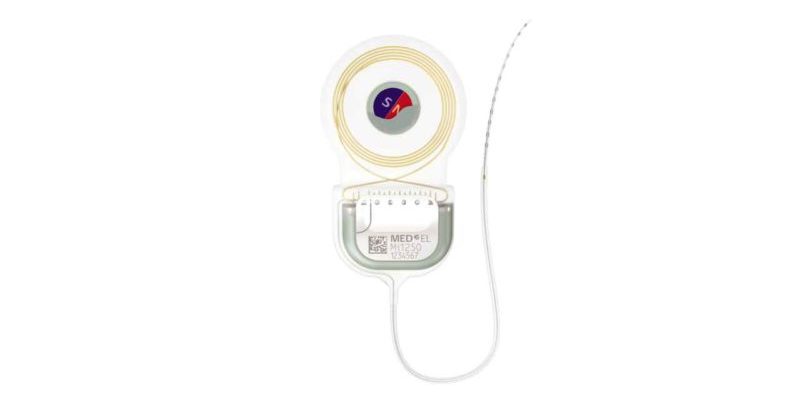
SYNCHRONY 2 cochlear implant with self-aligning magnet.
This leap forward in safe MRI access for cochlear implants isn’t just a “nice-to-have”; it can make a serious difference in comfort and safety for patients and can save time, effort, cost, and care for clinicians and healthcare systems.
This is what motivates us to always innovate: knowing that these improvements could have a significant impact on the lives of so many patients and clinicians.
Today we are excited to share a detailed discussion on the importance of MRI from Dr. Nancy Young, Professor of Otolaryngology at Northwestern and a member of Northwestern’s Knowles Hearing Center. She is a member of MED-EL’s US advisory board.
In 1991, Dr. Young performed the first cochlear implant of a child in the city of Chicago. Her CI program is based at Lurie Children’s Hospital of Chicago in Illinois, a tertiary care children’s hospital that is part of Northwestern University. They have a multidisciplinary team that includes surgeons, seven audiologists, five speech pathologists, a social worker, and an educator of the hearing impaired. The pediatric CI program she founded is now the largest in the US and she has personally performed over 2,000 cochlear implantations.
In this discussion, Dr. Young explains why MRI is so important for every candidate and why certain axial magnet designs have led to frequent complications, and she shares her very insightful recent research into MRI in children with SYNCHRONY cochlear implants.
Let’s turn over now to Dr. Young for her perspective on the importance of safe MRI access for cochlear implants.
Expert Perspective: Dr. Nancy Young
When I began doing CIs, MRIs were only done of the brain. MRI technology has advanced. Now they are routinely done of the body. MRI is used by orthopedic specialists to evaluate the spine and knee, cardiologists to evaluate the heart, and oncologists to evaluate the abdomen, to name just a few examples.
It is reasonable to assume that a child who receives a CI will at some point in their life be recommended an MRI.
Increased reliance upon MRI in medicine is well documented. MRI is now widely available and relied upon by specialists, primary care physicians, and emergency department physicians for diagnosis of a wide variety of conditions. In some cases, an alternative type of imaging may answer the question at hand, but in others, information from the MRI may be very important to diagnosis or management.
In terms of MRI specifically for children who are CI candidates, pre-surgical MRI is superior because it delineates both cochlear and eighth nerve anatomy. It is also very advantageous for children because it minimizes exposure to ionizing radiation which may cause complications later in life. An effort to minimize radiation from CT and other modalities has been ongoing in the US for several decades. It is best to evaluate the eighth nerve by obtaining detailed sagittal oblique views of the internal auditory canal that contains the seven and eight nerves.
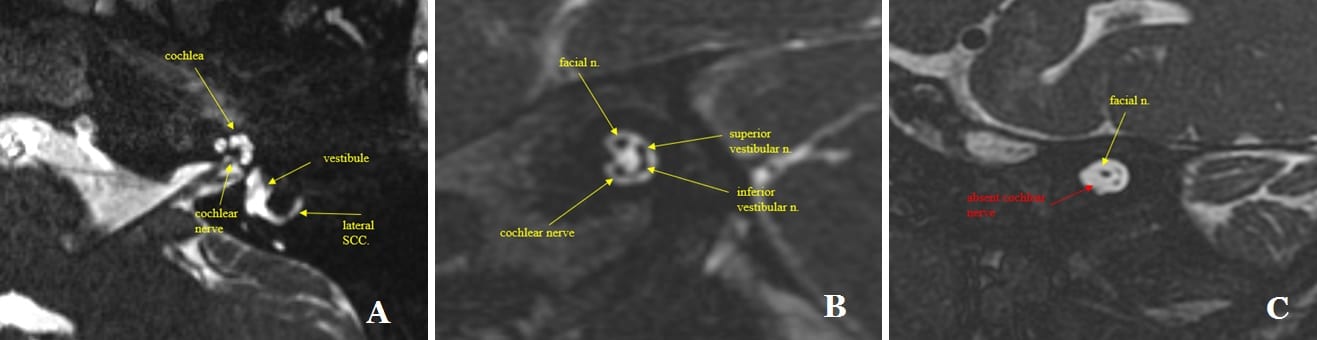
Pre-operative MRI assessment of cochlea and eighth nerve anatomy. A) Axial 3D T2 MRI image demonstrates normal appearance of the cochlea, lateral semicircular canal and the cochlear nerve. B) Sagittal oblique 3D MRI T2 image demonstrating the four nerve bundles in the internal auditory canal. The eight nerve divides into three branches: superior and inferior vestibular nerves and the cochlear nerve. Also present is the facial nerve. C) Sagittal oblique 3D T2 MRI image demonstrating hypoplasia of the eighth nerve in the internal auditory canal. The cochlear nerve is absent. The superior and inferior vestibular nerves are smaller than normal.
Pre-operative MRI can provide excellent images of the cochleae and has the advantage of identifying hypoplasia of the nerve of hearing which impacts pre-operative counseling, CI programming, rate of expected progress, and outcome. It also provides a baseline study of brain anatomy. At our center, we have identified brain abnormalities that required surgical or medical treatment pre-CI. Pre-CI MRI may also identify children who need brain MRI after implantation and may impact the choice of CI ear. For example, we recently identified an optic nerve tumor in an asymptomatic child undergoing CI evaluation. The child is receiving chemotherapy. The ear to be implanted will be opposite the affected eye to minimize artifact in future surveillance MRIs. This child will receive a SYNCHRONY device as we are confident the child will be able to undergo repeated MRI without complications based upon our experience.
Access to MRI after CI is important not only for elective studies that are scheduled well in advance, but for studies requested on an urgent basis. For example, MRI is now commonly used by emergency department physicians, often during hours when MRI administrative staff and CI team members are not readily available. Without easy MRI access, CT will often be substituted, which unnecessarily exposes children to ionizing radiation.
Challenges With Axial Magnets
When the first surgically removable axial magnet became available, the thought was that this would permit access to MRI for all CI patients. This is not what happened. The most important barrier to MRI for patients with removable axial magnets is patient resistance to even a relatively minor surgery. And, keep in mind it is two surgeries—one to remove and another to replace the axial magnet.
In addition, when the patient does agree to magnet removal, there is risk of tearing the soft silastic pocket when the magnet is removed and again when replaced. The pocket also becomes stretched with repeated removals/replacements, so it cannot be done more than a few times and still keep the magnet secure. Once the silastic pocket is damaged, reimplantation with a new device is necessary for appropriate retention of the transmitter coil or button-style processor.
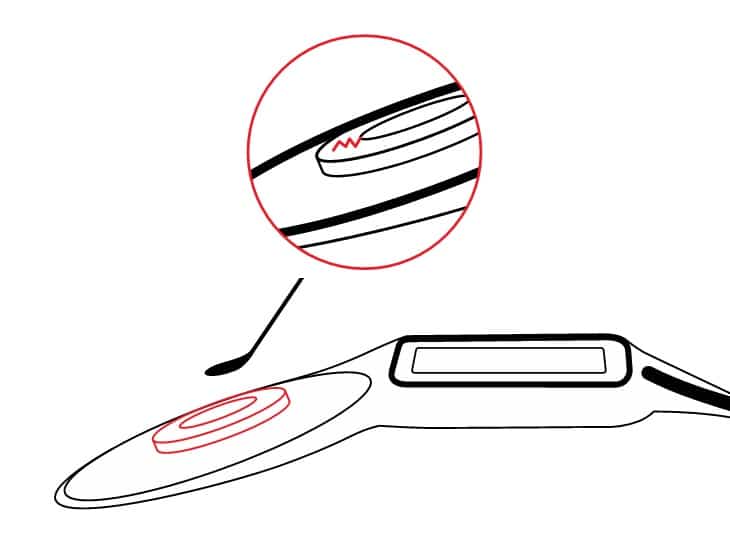
A tear in the soft silastic pocket can compromise magnet retention ability and may require full implant replacement.
So, a common outcome is the patient does not undergo the recommended MRI. Sometimes the medical issue for which MRI was desired is satisfactorily solved with alternative imaging methods. However, in other cases there is a delay in diagnosis or management with suboptimal information that may impact patient wellbeing.
We published a paper in Otology and Neurotology in 2016 about our experience with MRI of patients implanted with axial magnets: Magnetic Resonance Imaging of Cochlear Implant Recipients (Young et al. 2016).
When the first surgically removable axial magnet became available, the thought was that this would permit access to MRI for all CI patients. This is not what happened.
Complications related to the presence of the magnet occurred in 30% of episodes of imaging. The most common problem was rotation of the magnet. It may completely flip over and reverse polarity. One patient required surgery to replace the magnet because it slipped outside of the silastic pocket. Another experienced partial demagnetization. All of our patients with a magnet within their device underwent MRI under anesthesia.
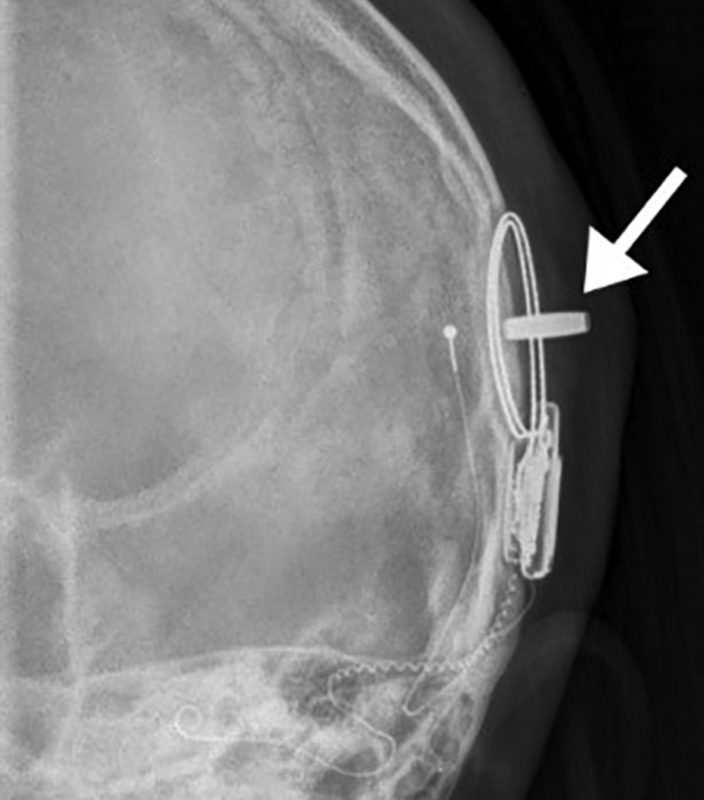
Skull Film X-ray done after MRI. The arrow is pointing to an axial magnet dislocated from the soft silastic pocket. The magnet is rotated 90 degrees causing pain and swelling. Surgery to replace it was required. Image credit: Young et al. 2020.
Requiring general anesthesia or sedation for all CI recipients undergoing MRI had been our protocol because of outward forces on the axial magnet causing discomfort or pain, which may provoke great anxiety. There are multiple published reports of adult CI recipients not completing MRI studies due to pain and anxiety. I am pleased to say that for our SYNCHRONY patients, the protocol has changed. We no longer require anesthesia or sedation unless needed for reasons other than the presence of CI.
One of the most interesting findings in our earlier study of devices with an axial magnet was that all magnet complications occurred during body studies. The forces on the magnet are more likely to be greater during body or spine studies, depending upon multiple factors including the height of the patient. The increased likelihood of problems during studies below the head and neck was not recognized before in the literature.
Prior to the introduction of the MEDEL SYNCHRONY in 2015, all MRI of CI recipients were scheduled with sedation or general anesthesia at our hospital. We have a CI audiologist perform the head wrap after the child is asleep in the MRI department. We do this because the audiologist understands where the receiver-stimulator and magnet within it are located. The result is a more effective headwrap than if the MRI staff places it. However, this further complicates scheduling. More important is that having a headwrap does not eliminate discomfort or magnet movement for many patients with axial magnets.
In our experience, obtaining MRI for patients who have devices with removable axial magnets is challenging and does not lend itself to urgent MRI. Because of the discomfort generated by forces on the magnet inside of the MRI machine, kids with axial magnets need sedation or general anesthesia, and a head wrap. Much coordination is needed for this to occur.
No patient with a CI at our hospital had ever undergone an awake emergent MRI requested by the emergency department until we began to use the MED-EL SYNCHRONY with diametric magnet.
SYNCHRONY Advantage in MRI
I have been using the SYNCHRONY since it was commercially approved by the FDA in 2015. We strongly recommended it to all patients for whom we know future MRIs will be necessary. I like the design of the space that contains the SYNCHRONY’s diametric magnet. It is housed in the undersurface of the device. So, it is well protected from external trauma.
In our experience, the diametric magnet housed on the undersurface of the SYNCHRONY solves the magnet movement/displacement/pain/anxiety issues reported in the literature. No headwrap makes the workflow much easier.
Prior to the introduction of the MEDEL SYNCHRONY in 2015, all MRI of CI recipients were scheduled with sedation or general anesthesia at our hospital.
I have not yet had to remove a SYNCHRONY magnet for any reason. I am very glad to learn that MED-EL’s bench testing reveals that many removals and replacements may be done without damage, as this will be an important long-term advantage for some recipients, including those whose future need for serial MRI is unknown at time of implantation.
Clinical Research: MRI with SYNCHRONY in Children
In 2020, we published our MRI experience with the SYNCHRONY with a diametric magnet in place. This publication is available open access in Laryngoscope: Impact of Cochlear Implant With Diametric Magnet on Imaging Access, Safety, and Clinical Care. Young et al. 2020.
Seven children between ages 15 months and 19 years (mean of 8.4 years) underwent MRI. These seven patients underwent 17 sessions of MRI during which 18 different MRI studies were obtained. There were 14 brain and four body or spine studies. The studies were done for a variety of reasons including evaluation of brain tumor reoccurrence, evaluation of brain ventricle size to rule out hydrocephalus, and follow-up after drainage of a spine abscess. No child received a headwrap prior to MRI.
Five of the brain studies were “fast brain” studies to evaluate ventricular size. A fast study is typically done to evaluate ventricular size. It was developed to avoid repeated CT scans with ionizing radiation that children with hydrocephalus requiring a shunt frequently required. This type of MRI had never been done in any of our CI recipients because it is such a short study (5-6 minutes) that it did not make sense to put them under general anesthesia to obtain a brief limited study instead of a complete brain study. Because the SYNCHRONY enables awake MRI, the fast brain study became an option for our CI patients, including a 21-month-old. And, one patient in the emergency department successfully had a fast brain study to rule out acute hydrocephalus. This was a milestone event at our hospital for a cochlear implant recipient.
Two patients underwent MRI only 28 days after CI surgery without problems, including one with bilateral SYNCHRONY devices. Another patient underwent awake MRI in the stronger 3T MRI scanner twice without incident.
Of the 17 MRI sessions, 13 were done awake and four were done under anesthesia due to patient age and ability to cooperate, not due to presence of the CI. Remarkably, there were no reports of pain or discomfort during or after MRI. No magnet movement or other magnet complications occurred. No urgent calls to CI team members regarding these patients occurred, which was a welcome change.
Remarkably, there were no reports of pain or discomfort during or after MRI. No magnet movement or other magnet complications occurred. No urgent calls to CI team members regarding these patients occurred, which was a welcome change.
We also evaluated the diagnostic usefulness of the MRI scans obtained. Artifact was expected for brain studies, but the question we asked was whether the study was able to answer the question being asked, in other words, was it diagnostically useful.
All but one of the 18 studies met the goal of diagnostic usefulness. This included 13 of 14 (93%) of brain studies. This latter finding is in line with our prior experience. Imaging of the brain after CI is usually successful when only one implant is present. The presence of two implants does create significant artifact that may limit imaging. Depending on the clinical problem and the location of the area of interest, we may not do bilateral CI for some patients, such as those requiring tumor surveillance.
Further Research
I am an advocate for early cochlear implantation. Improved language outcome of infant implantation has been demonstrated, perhaps most compellingly by investigators at the University of Melbourne in Australia. In 2019 we published a study highlighting the safety of implantation of infants and young children using modern anesthesia and surgical techniques: Safety and Effectiveness of Cochlear Implantation of Young Children, Including Those With Complicating Conditions (Hoff et al. 2019).
Our CI team is also leading MED-EL’s multicenter FDA clinical trial to expand pediatric indications. Expanding age of implantation to include infants as young as 7 months of age is important to improve access to CI for US children. I am grateful to MEDEL for their leadership in launching the first FDA clinical trial in many years to expand pediatric indications.
Our research group is focused on improving pediatric CI outcomes by using pre-surgical MRI of the brain to predict language. The goal of this research is to determine the type and dose of behavioral therapy necessary to optimize outcomesfor each child—precision therapy, analogous to precision medicine. Our research involves the use of machine learning. We have a series of young implanted patients in our proof of concept paper published open access in The Proceedings of the National Academy of Sciences in 2018: Neural preservation underlies speech improvement from auditory deprivation in young cochlear implant recipients (Feng et al. 2018). Our research in this area is supported by NIH/National Institute of Deafness and Other Communication Disorders: R21DC016069-01A1.
Thank you Dr. Young for sharing your expert insight into MRI with cochlear implants!
Great Protection. Guaranteed.
Because of our long and positive experience with MRIs and cochlear implants, we’re the only hearing implant company to offer a life-long MRI guarantee. We promise to replace the implant in the very unlikely event that it’s damaged during an MRI scan.*
- Valid for all MED-EL multichannel cochlear implants since 1994
- Life-long and worldwide
- The first and only to be offered by any hearing implant producer
* The terms and conditions of the MRI Guarantee can be found here.
** MED-EL cochlear implants since 1994 are MR conditional. Recipients with a MED-EL cochlear implant may be safely MRI scanned following the conditions detailed in the instructions for use.
References

Nancy M. Young, MD, FACS, FAAP
Nancy Young, MD, founded the Lurie Children’s Cochlear Implant Program and is Medical Director of Audiology. She is fellowship-trained in Neurotology (Advanced Ear Surgery). Dr. Young edited “Pediatric Cochlear Implantation: Learning and the Brain” published in 2016. She was recently elected to the American Otological Society (AOS) Council, the leadership body of the second oldest medical society in the U.S.
Was this article helpful?
Thanks for your feedback.
Sign up for newsletter below for more.
Thanks for your feedback.
Please leave your message below.
CTA Form Success Message
Send us a message
Field is required
John Doe
Field is required
name@mail.com
Field is required
What do you think?
The content on this website is for general informational purposes only and should not be taken as medical advice. Please contact your doctor or hearing specialist to learn what type of hearing solution is suitable for your specific needs. Not all products, features, or indications shown are approved in all countries.

Nancy M. Young, MD, FACS, FAAP
Nancy Young, MD, founded the Lurie Children’s Cochlear Implant Program and is Medical Director of Audiology. She is fellowship-trained in Neurotology (Advanced Ear Surgery). Dr. Young edited “Pediatric Cochlear Implantation: Learning and the Brain” published in 2016. She was recently elected to the American Otological Society (AOS) Council, the leadership body of the second oldest medical society in the U.S.

Nancy M. Young, MD, FACS, FAAP
Nancy Young, MD, founded the Lurie Children’s Cochlear Implant Program and is Medical Director of Audiology. She is fellowship-trained in Neurotology (Advanced Ear Surgery). Dr. Young edited “Pediatric Cochlear Implantation: Learning and the Brain” published in 2016. She was recently elected to the American Otological Society (AOS) Council, the leadership body of the second oldest medical society in the U.S.



